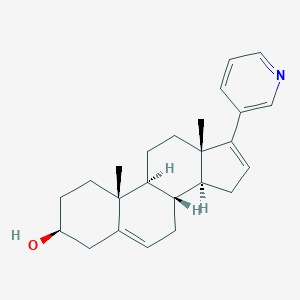
Abiraterone
Please note, this OEL/ADE monograph also applies to abiraterone acetate (CAS RN 154229-18-2). Abiraterone is a drug used in castration-resistant prostate cancer (formerly hormone-resistant or hormone-refractory prostate cancer) — i.e., prostate cancer not responding to androgen deprivation or treatment with antiandrogens. It is formulated as the prodrug abiraterone acetate.
Why do you need a Abiraterone monograph?
Superior Toxicology and Wellness’s monographs are a cost-effective and convenient way to meet the requirements for PDEs (ADEs)
- The numerical OEL and control band assignment needed to determine the level of required engineering controls and personal protective equipment
- A listing of all cited references utilized in the derivation of the OEL and ADE
- An expert review and discussion with respect to the critical endpoints of concern, the rationale for the choice of endpoints, and dose that is to be used in the derivation of the ADE (PDE), as required by the EMA’s Guideline on setting health-based exposure limits for use in risk identification in the manufacture of different medicinal products in shared facilities
Benefits of Superior Toxicology and Wellness monographs:
- Full drug documentation. The report consists of 10-12 pages in length, with calculations and cited references.
- Quick delivery! 24-48 hours of purchase. Sometimes even an instant download.
- Save money. Similar reports will cost you potentially 5-12 times more.
- Search our other reports!

Reviews
There are no reviews yet.